| Producent |
|---|
Ortopedia
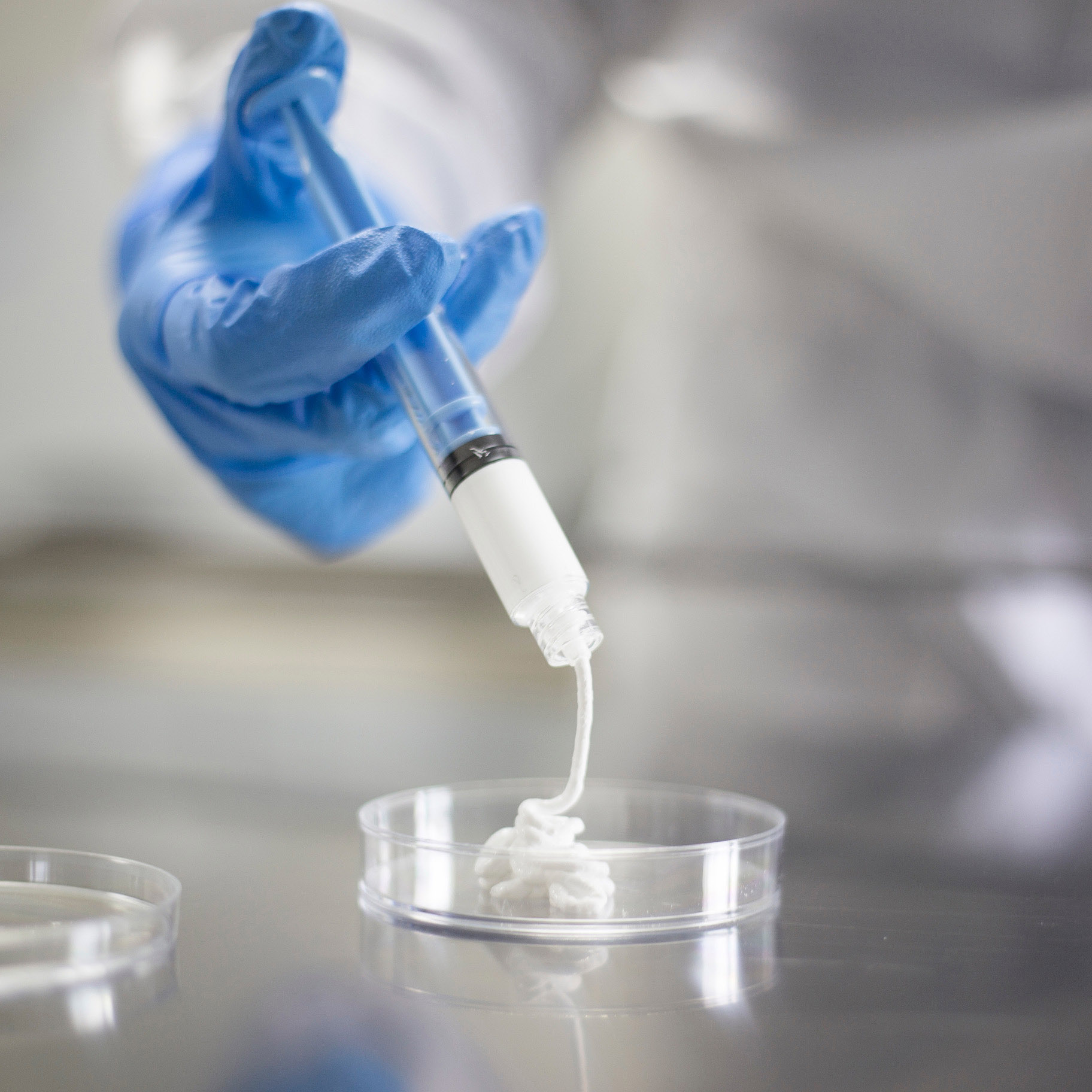
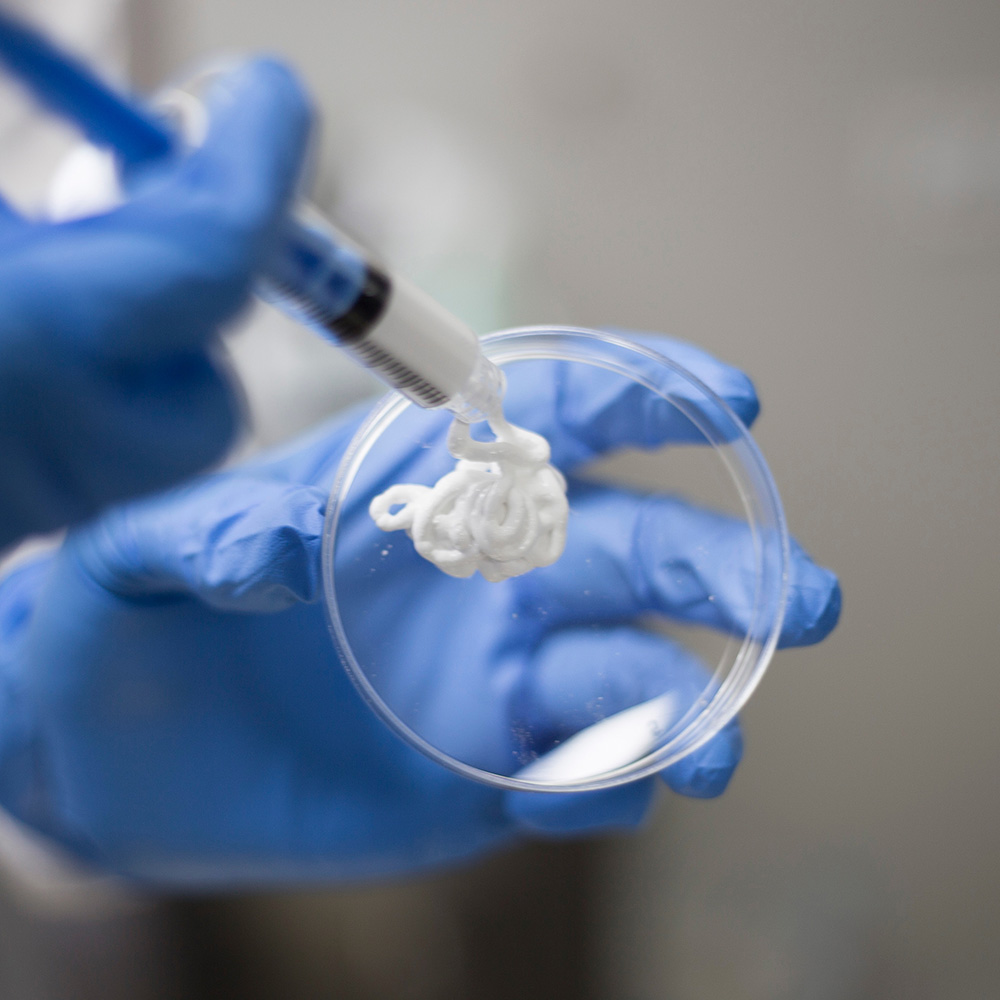
Biongraft – syntetyczny substytut kostny
Substytut kostny zbudowany z beta-TCP oraz tlenku cyrkonu (ZrO2) o właściwościach antybakteryjnych.
Produkt resorbowalny, o właściwościach osteokonduktywnych, zastępowany kością podczas procesu gojenia.
Dostępny pod postacią: granulek, pasty, bloczków, klinów.
Zastosowanie: trauma, rewizje, osteotomie, wypełnianie pustych przestrzeni w chirurgii kręgosłupa.
Podobne produkty
Genex® – substytut kości w postaci pasty
Ortopedia