Med&Care has partnered with Biocomposites to distribute STIMULAN. Approved for use directly at the site of infection in bone and soft tissue. Mixed with antibiotics to protect the implant from colonization by bacteria.
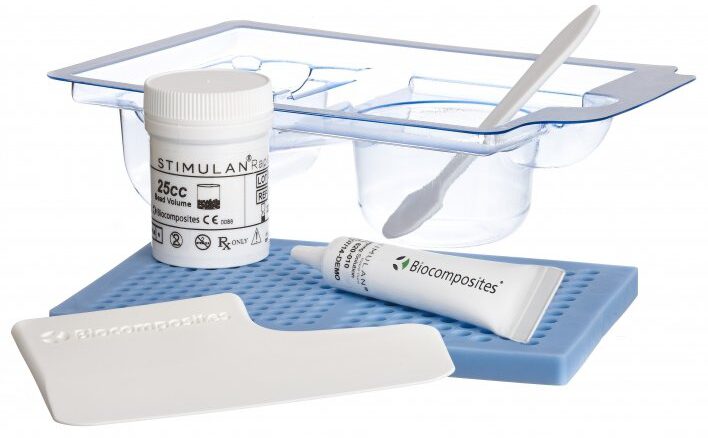
Med&Care has partnered with Biocomposites to distribute STIMULAN. Approved for use directly at the site of infection in bone and soft tissue. Mixed with antibiotics to protect the implant from colonization by bacteria.
